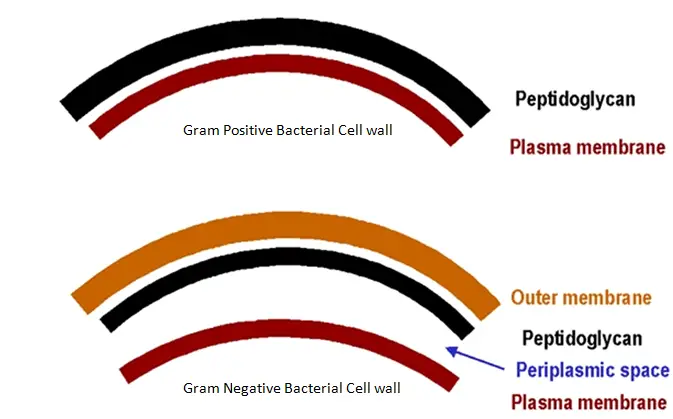
The influence of bacteria on host cells is mediated by bacterial antigens and structural components (modulins), as well as lipopolysaccharides (LPS or endotoxins), peptidoglycans, lipoteichoic acids (LTA), fimbriae, flagella, outer membrane proteins, and exopolysaccharides, which altogether comprise MAMPs and determine bacterial virulence ( J. Certainly, regardless of bacteria’s pathogenicity to the host, all bacteria produce microbe-associated molecular patterns (MAMPs) or pathogen-associated molecular patterns (PAMPs) that are recognizable to the host cell pattern-recognition receptors (PRRs) ( Zhong et al., 2017). Noteworthy, residual bacterial biofilms and/or their components can persist in dentine tubules even after RET disinfection protocols and could potentially influence RET outcomes ( Verma et al., 2017). The microbiota of infected root canals housed in closed environments is comprised of bacteria that belong to the phyla Bacillota, Bacteroidota, Fusobacteriota, Actinomycetota, and Pseudomonadota ( Sakamoto et al., 2006 Siqueira et al., 2011 Manoharan et al., 2020) with a high prevalence of anaerobic species of the genus Bacteroides, Corynebacterium, Peptostreptococcus, and Fusobacterium ( Bergenholtz, 1974 Sundqvist and Figdor, 2003). Although promising, the successful outcome of RET has been jeopardized by bacterial persistence in the root canal, affecting the viability, proliferation, immune response, and differentiation potential of stem cells from the apical papilla (SCAP) ( Petridis et al., 2018a). Regenerative endodontic treatment (RET), a biological-based procedure, has shown promising clinical results evidencing root development ( Wikström et al., 2022) and pulp tissue regeneration ( Xuan et al., 2018). Indeed, in young children with immature teeth, infection will lead to the death of odontoblast cells and consequent interruption of root maturation ( Hecova et al., 2010). In this regard, polymicrobial oral microflora can invade normally sterile tissues like dental pulp and cause severe tooth damage. Violations of the integrity of the tooth by caries or traumatic dental injuries (TDI) increases the risk of developing pulp tissue inflammation, pulp necrosis, and periapical bone destruction ( Fouad, 2019). SCAP’s secretome assessed with Multiplex ELISA showed the upregulation of pro-inflammatory factors IL-6, IL-8, GM-CSF, IL-1b, neurotrophic factor BDNF, and angiogenic factor VEGF, induced by UV-C–killed bacteria.ĭiscussion: The results suggest that long term stimulation (for 21 days) of SCAP with UV-C–inactivated bacteria stimulate their mineralization and inflammatory response, while DNA influence has no such effect, which opens up new ideas about the nature of RET failure. coli lipopolysaccharide (LPS) positive control in the case of UV-C–inactivated bacteria meanwhile, DNA treatment decreased the level of ALP compared to the positive control. Immunofluorescence analysis detected that the early mineralization marker alkaline phosphatase (ALP) was increased by the level of E. Results: We showed that mineralization promotion was greater with UV C–inactivated bacteria compared to bacterial DNA. Histochemical staining using Osteoimage Mineralization Assay and Alizarin Red analysis was performed to study SCAP mineralization, while inflammatory and osteo/odontogenic-related responses of SCAPs were assessed with Multiplex ELISA. :max_bytes(150000):strip_icc()/gram_positive_vs_negative-5b7f26d2c9e77c005746fbd7.jpg)
Methods: To assess the effect of bacterial remnants on SCAP, we used UV-C–inactivated bacteria (as cell wall-associated virulence factors) and bacterial DNA. The aim of the study was to investigate whether prolonged exposure of stem cells from the apical papilla (SCAP) to bacterial remnants of Fusobacterium nucleatum, Actinomyces gerensceriae, Slackia exigua, Enterococcus faecalis, Peptostreptococcaceae yurii, commonly found in infected traumatized root canals, and the probiotic bacteria Lactobacillus gasseri and Limosilactobacillus reuteri, can alter SCAP’s inflammatory response and mineralization potential. This interference is claimed to be caused by the interaction of bacteria that reside in the root canal with the stem cells that are one of the essentials for RET. Introduction: Bacterial persistence is considered one of the main causal factors for regenerative endodontic treatment (RET) failure in immature permanent teeth. 5Wallenberg Centre for Molecular Medicine, Umeå University, Umeå, Sweden.4Section for Anatomy, Department of Integrative Medical Biology (IMB), Umeå University, Umeå, Sweden. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
